Meet USDM at Glean:GO 2026.
Glean:GO brings together customers, partners, AI leaders, IT teams, digital workplace leaders, and business executives shaping how work gets done with enterprise AI.
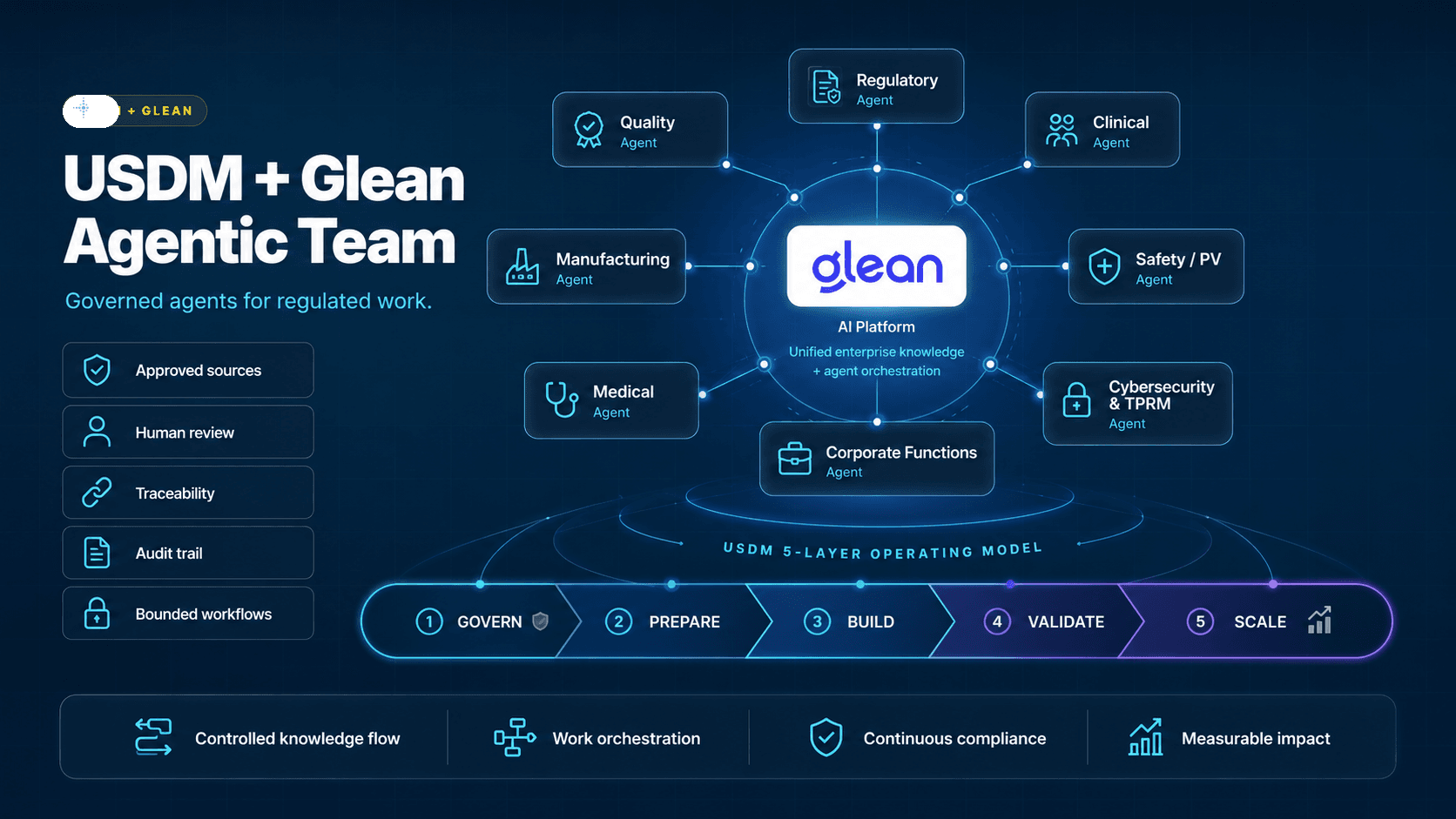
USDM is sponsoring the event to help life sciences teams understand how governed Work AI can connect enterprise knowledge, respect permissions, support traceability, and move regulated organizations from AI experimentation to measurable business impact.
Why attend
Enterprise AI, through a regulated lens
Glean:GO focuses on the architecture, strategies, and practical skills needed to power the next generation of enterprise AI. USDM brings the life sciences compliance, validation, and adoption perspective to that conversation.
Knowledge-first AI for life sciences
Glean connects enterprise knowledge across systems. USDM helps regulated organizations turn that capability into governed workflows for Quality, Regulatory, Clinical, Manufacturing, Commercial, and IT teams.
From AI intent to AI impact
The opportunity is not another chatbot demo. It is permission-aware knowledge access, practical agents, validated workflows, and an operating model that life sciences teams can defend.
What to talk with USDM about at Glean:GO
- AI readiness and governance for regulated life sciences organizations.
- Glean implementation patterns for permission-aware enterprise search, answers, and agents.
- Use cases including inspection readiness, SOP impact analysis, evidence retrieval, quality document review, and regulatory knowledge workflows.
- Validation strategy, risk assessment, traceability, and controls for AI-enabled workflows.
- Adoption programs that help teams use Work AI safely and consistently across the enterprise.
About USDM and Glean
Glean’s Work AI platform brings enterprise search, AI answers, agents, and connectors into a unified experience designed to respect existing permissions and make enterprise knowledge easier to use.
USDM helps life sciences organizations deploy Glean in regulated environments with GxP implementation discipline, validation strategy, governance, adoption, and life sciences-specific agents and connectors.